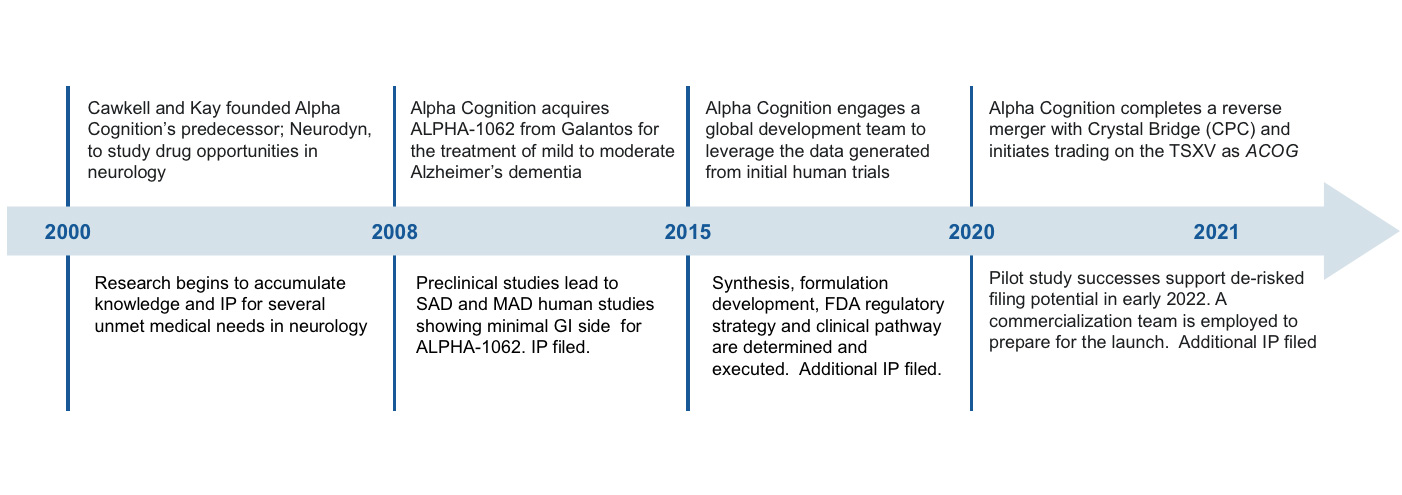
Alpha Cognition is a clinical stage, biopharmaceutical company dedicated to developing treatments for patients suffering from neurodegenerative diseases, such as Alzheimer's disease, and Cognitive Impairment with Mild Traumatic Brain Injury (mTBI) for which there are limited or no treatment options. The Company is focused on the development of ALPHA-1062 for the treatment of mild-to-moderate Alzheimer’s disease and received an accepted NDA from FDA in December 2023. The company anticipates an FDA approval in mid-2024. The Company’s ALPHA-1062 development program is primarily focused on clinical and regulatory development, Chemistry, Manufacturing and Control (CMC) development, and commercial readiness. The Company has three additional development programs: ALPHA-1062 in combination with memantine for the treatment of moderate-to-severe Alzheimer’s disease, ALPHA-1062 intranasal formulation for the treatment of cognitive impairment with mTBI, and ALPHA-0602, previously referred to as ‘Progranulin,’ for the treatment of amyotrophic lateral sclerosis, otherwise known as ALS or Lou Gehrig’s disease.
ALPHA-1062 is a patented new chemical entity being developed as a next generation acetylcholinesterase inhibitor (AChEI) for the treatment of Alzheimer’s disease, with expected minimal gastrointestinal side effects. ALPHA-1062’s active metabolite is differentiated from donepezil and rivastigmine in that it binds neuronal nicotinic receptors, most notably the alpha-7 subtype, which is known to have a positive effect on cognition. ALPHA-1062 is in development in combination with memantine to treat moderate-to-severe Alzheimer’s disease and as an intranasal formulation for cognitive impairment with mTBI.
ALPHA-0602 (Progranulin) is expressed in several cell types in the central nervous system and in peripheral tissues, promotes cell survival, regulates certain inflammatory processes, and plays a significant role in regulating lysosomal function and microglial responses to disease. Its intended use for the treatment of neurodegenerative diseases has been patented by the Company and ALPHA-0602 has been granted an Orphan Drug Designation for the treatment of ALS by the FDA. ALPHA-0702 and ALPHA-0802 are Granulin Epithelin Motifs, (“GEMs”), derived from full length progranulin which have therapeutic potential across multiple neurodegenerative diseases. GEMs have been shown to be important in regulating cell growth, survival, repair, and inflammation. ALPHA-0702 and ALPHA-0802 are designed to deliver this with potentially lower toxicity, and greater therapeutic effect.