Alpha Cognition Announces Third Quarter and Nine Months Ended September 30, 2023, Results and Provides Corporate Update
November 28, 2023
VANCOUVER, B.C., November 28, 2023. Alpha Cognition Inc. (CSE: ACOG) (OTCQB: ACOGF) (“Alpha Cognition”, or the “Company”), a biopharmaceutical company committed to developing novel therapies with the potential to transform the lives of people with debilitating neurodegenerative disorders, today reported financial results for the third quarter and nine months ended September 30, 2023, and provided a corporate update.
"We recently achieved a significant milestone in our journey with the submission of our New Drug Application to the FDA at the end of September. Our dedicated team has tirelessly advanced our lead product, and we now stand on the cusp of a new chapter, confident in the strength of our data and the expertise that underpins our submission. The Company expects FDA approval in the second half of 2024. We believe that ALPHA-1062 will offer a meaningful differentiated therapy for patients with Alzheimer’s dementia" said Michael McFadden, the Company’s Chief Executive Officer.
Corporate Updates
NDA Submission
Alpha Cognition announced on September 29, 2023, the submission of a New Drug Application (“NDA”) to the U.S. Food and Drug Administration (“FDA”) for ALPHA-1062, a proprietary, patented, delayed release oral tablet formulation in development for the treatment of mild-to-moderate Alzheimer’s Disease. This submission marks a significant milestone in the company's ongoing commitment to advancing patient care for patients suffering from Alzheimer’s disease.
Issued Patent
Alpha Cognition received from the US patent office issued patent No. 11,795,176 titled “Solid Forms of ALPHA-1062 Gluconate” which includes claims for crystalline solid forms of ALPHA-1062 and will bolster existing patents that the company holds. The Company expects to file additional patents for ALPHA-1062 within the quarter, which will build on the robust patent portfolio the company already holds.
Awarded Department of Defense $750,000 R&D Grant
On June 5, 2023, the Company was awarded a $750,000 research and development grant from the Army Medical Research and Material Command for a pre-clinical study on the use of the ALPHA-1062IN to reduce blast mTBI (mild Traumatic Brain Injury) induced functional deficit and brain abnormalities. The study grant is issued in collaboration with the Seattle Institute of Biomedical and Clinical Research and is endorsed by the Department of Defense. Researchers initiated the study in Q3 2023 and the Company expects to report initial findings in Q2, 2024.
PPM Financing
On May 30, 2023, the Company announced a best-efforts private placement offering to raise gross proceeds of up to US$6.5 million of units at a price of $0.22 per unit. Each unit consists of one common share and one-half of a common share purchase warrant. Each whole warrant will entitle the holder to purchase an additional common share of the Company at a price of US$0.31 per share for a period of three years from the closing date. The Company expects to use the net proceeds from the offering for research and development, general and administrative matters, and working capital. The Company has US$3.8 million remaining of the US$6.5 million of units to be issued in the private placement, as of the date of this press release.
TBI Out-Licensing Plan
The Company continued its work to out-license to a newly formed private entity, Alpha Seven Therapeutics, Inc. (“Alpha Seven”) which will develop ALPHA-1062Intranasal (IN) for applications in traumatic brain injury. Alpha Seven will focus on the advancement of ALPHA-1062IN initially for the treatment of Cognitive Impairment with mTBI using an intra-nasal formulation, including advancing clinical trials with the goal of FDA approval. The establishment of Alpha Seven Therapeutics is expected to provide for separate funding and advancement of the TBI applications of ALPHA-1062IN while permitting Alpha Cognition to remain focused on advancing ALPHA-1062 for use in the treatment of symptoms of Alzheimer’s disease. If successful, the Alpha Seven’s advancement of this program in 2023 will allow for potential upcoming inflection points including IND acceptance and first patient enrolled in Ph2 trial during 2H 2024, followed by initial Phase 2 clinical results.
Financial Highlights for Second Quarter and Nine months ended September 30, 2023
(Expressed in United States Dollars)
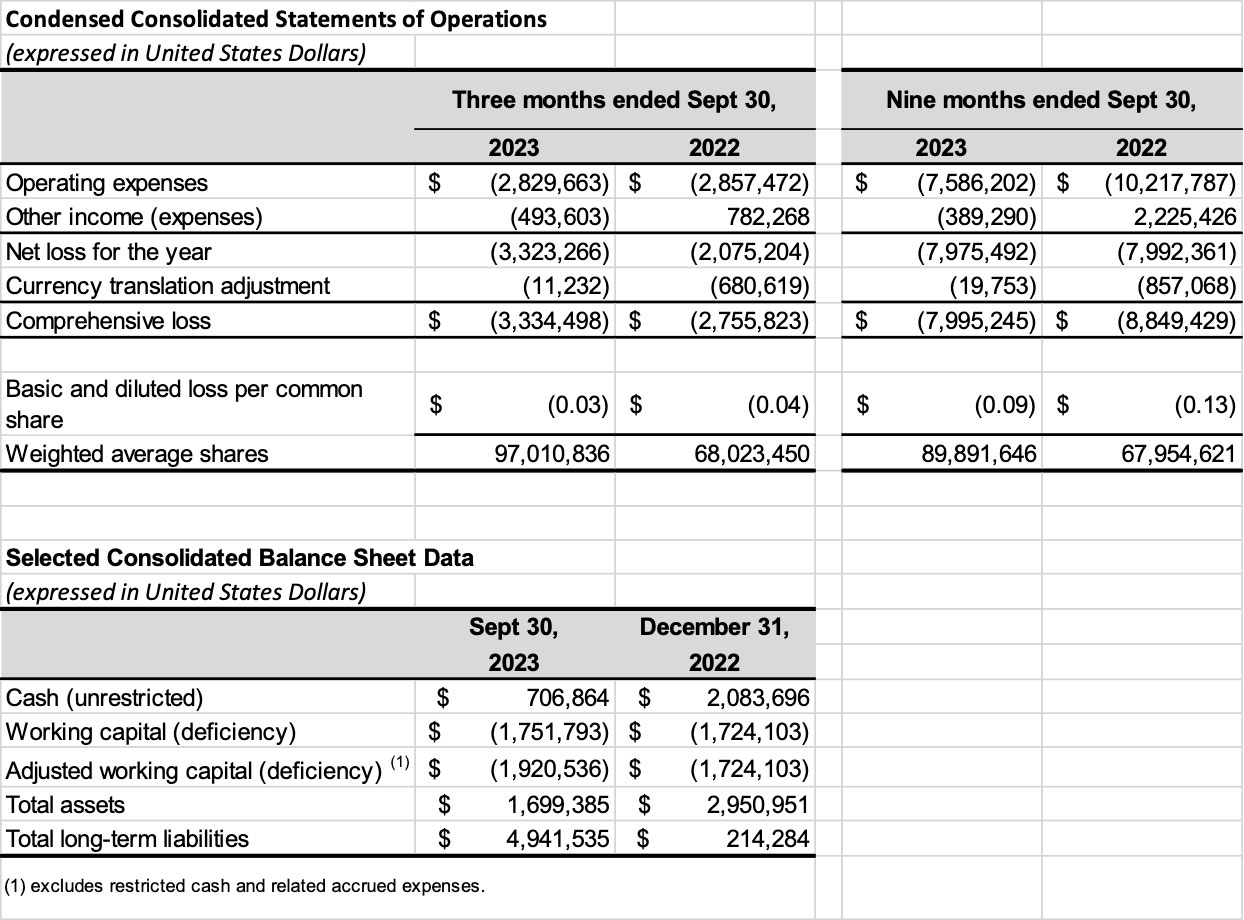
- Research and development (R&D) expenses were $1.5 million for the three months ended September 30, 2023, and $3.9 million for the nine months ended September 30, 2023, compared to $1.6 million and $6.4 million in the same periods in 2022, respectively. R&D expenses decreased from the prior year primarily due to the completion during 2022 of the main clinical trails for ALPHA-1062.
- General and administrative (G&A), excluding non-cash expenses relating to accretion, amortization, depreciation, and share-based compensation, were $0.8 million for the three months ended September 30, 2023, and $2.2 million for the nine months ended September 30, 2023, compared to from $1.0 million and $2.8 million in the same periods of 2022 respectively. The G&A expense decreases for both the three and nine months ended September 30, 2023 was primarily related to lower management fees and salaries, investor relations expense, professional fees, financing costs, and subcontractors expenses, offset some by higher consulting fees. The overall G&A decrease year over year was due to the Company’s cost cutting efforts and focus on the advancement if its main asset, ALPHA-1062 in AD.
- The Company recorded a loss on revaluation derivative liability for the three months ended September 30, 2023, of $0.5 million and $0.5 million for nine months ended September 30, 2023, compared to a gain of $0.3 million and $1.7 million in the same periods of 2022 respectively. On August 31, 2023, the Company’s functional currency changed to USD from CAD; as such, the Company recorded a derivative liability on the warrants outstanding with previously issued CAD exercises prices and the derivative liability on warrants previously issued in USD was derecognized. The Company performs a revaluation each reporting period for its derivative liability and records the change in fair value of its warrants issued in a currency that differs from the functional currency at the end of each period.
- Effective August 31, 2023, the functional currency of the Company was updated to USD as management assessed that the currency of the primary economic environment in which the Company operates changed to USD on that date. The Company reclassified its derivative liabilities upon the change in functional currency, which resulted in an increase of $4,189,576 in derivative liabilities with a corresponding increase in reserves of $351,969 and decrease in share capital of $4,541,545 at August 31, 2023.
- Share-based compensation under general and administrative was $0.5 million for the three months ended September 30, 2023, and $1.5 million for nine months ended September 30, 2023, compared to $0.2 million and $1.0 million in the same periods of 2022, respectively. The 2023 increases was primarily related to new stock option grants issued in 2023, the repricing of previously issued stock options during the first quarter of 2023, and related fluctuations in the Company’s stock price over the periods.
- The Company incurred foreign exchange gain/losses of nil in the three months ended September 30, 2023, and nil for the full nine months ended September 30, 2023, compared to a gain of $0.5 million and $0.5 million in the same periods of 2022, respectably.
- The third quarter of 2023 net loss was $3.3 million, or a net loss of $0.03 per share, and for the nine months ended September 30, 2023, net loss was $8.0 million, or a net loss of $0.09 per share, compared to the third quarter of 2022 net loss of $2.8 million, or a net loss of $0.04 per share, and for the nine months ended September 30, 2023, net loss of $8.8 million, or a net loss of $0.13 per share.
- Cash and cash equivalents at September 30, 2023 were $0.9 million, including $0.2 million in restricted cash.
- Shares of common stock outstanding at September 30, 2023 were 101,064,722.
About Alpha Cognition Inc.
Alpha Cognition Inc. is a clinical stage, biopharmaceutical company dedicated to developing treatments for patients suffering from neurodegenerative diseases, such as Alzheimer’s disease and Cognitive Impairment with mild Traumatic Brain Injury (“mTBI”), for which there are currently no approved treatment options.
ALPHA-1062, is a patented new chemical entity being developed as a new generation acetylcholinesterase inhibitor for the treatment of Alzheimer’s disease, with expected minimal gastrointestinal side effects. ALPHA-1062’s active metabolite is differentiated from donepezil and rivastigmine in that it binds neuronal nicotinic receptors, most notably the alpha-7 subtype, which is known to have a positive effect on cognition. ALPHA-1062 is also being developed in combination with memantine to treat moderate to severe Alzheimer’s dementia, and as an intranasal formulation for Cognitive Impairment with mTBI.
For further information:
Michael McFadden, CEO
Tel: 1-858-344-4375
info@alphacognition.com
https://www.alphacognition.com/
Neither Canadian Securities Exchange (the “CSE”) or the OTC Markets Group, accepts responsibility for the adequacy or accuracy of this release.
Forward-looking Statements
This news release includes forward-looking statements within the meaning of applicable securities laws. Except for statements of historical fact, any information contained in this news release may be a forward‐looking statement that reflects the Company’s current views about future events and are subject to known and unknown risks, uncertainties, assumptions and other factors that may cause the actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. In some cases, you can identify forward‐looking statements by the words “may,” “might,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “objective,” “anticipate,” “believe,” “estimate,” “predict,” “project,” “potential,” “target,” “seek,” “contemplate,” “continue” and “ongoing,” or the negative of these terms, or other comparable terminology intended to identify statements about the future. Forward‐looking statements may include statements regarding the TBI out-licensing plan and associated financing, the availability of funding pursuant to financings, the Company’s business strategy, market size, potential growth opportunities, capital requirements, clinical development activities, the timing and results of clinical trials, regulatory submissions, potential regulatory approval and commercialization of the Company’s products. Although the Company believes to have a reasonable basis for each forward-looking statement, we caution you that these statements are based on a combination of facts and factors currently known by us and our expectations of the future, about which we cannot be certain. The Company cannot assure that the actual results will be consistent with these forward-looking statements. These forward‐looking statements speak only as of the date of this news release and the Company undertakes no obligation to revise or update any forward‐looking statements for any reason, even if new information becomes available in the future.